 Polymers were dissolved in deionised water on a shaker for at least 72 h. Solution parameters like density, viscosity, surface tension and conductivity were measured to investigate their influence on the electrospinning process. Sorbitan trioleate (Span85, Sigma-Aldrich) was added for adjusting surface tension. Different concentrations and blends were tested (see Table 1). Heparin (5000 IE/ml, Biochrom) was added to avoid coagulation. The lyophilised porcine plasma (PPlas) and PEO (400 kDa, Sigma-Aldrich) were dissolved in deionised water on a shaker for at least 72 h. Biopolymer solutions were blended with non-toxic synthetic and water-soluble polyethylene oxide (PEO) to improve spinnability. Blood protein powder was created by lyophilisation. Cell count (Advia120, Siemens) and protein concentration (Pierce BCA Protein Assay Kit, Thermo Fisher Scientific) were measured to characterise resulting blood plasma. 2 Material and methods 2.1 Biopolymer solution for electrospinningĪnti-coagulated porcine blood from the slaughterhouse was centrifuged with ≈ 2000 rcf (relative centrifugal force, Heraeus Megafuge 1 S-R, Thermo Fisher Scientific) for 10 min to separate blood components. In addition, cross-linking with glutaraldehyde vapour will be performed and its influence on long-term biostability has to be analysed. The aim of this study is to develop an electrospinning setup for the fabrication of blood protein based patient-specific scaffolds for cardiovascular applications. A disadvantage of choosing electrospun protein based scaffolds is their high hydrolytic degradation rate in a physiological environment. The biodegradable scaffolds will be gradually replaced by infiltrating and proliferating cells, leading to an effective tissue remodelling. 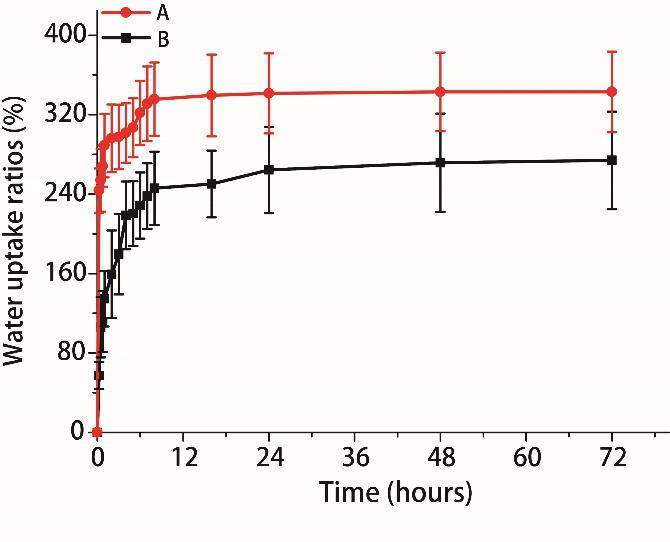
To reduce undesired immune reactions for patients, the use of biopolymer solutions from blood proteins is desirable because proteins contain specific structural and functional information and are biodegradable. Electrospinning is a promising method for the fabrication of biocompatible fine-fibres and porous scaffolds. Therefore, it is important to develop biocompatible scaffolds for cardiovascular applications that have no undesirable effects on the surrounding tissue. The aim of tissue engineering is a functional tissue replacement by culturing cells on suitable scaffolds. Therefore, the development of a suitable material for the replacement of diseased vascular tissue is desirable. However, the use of such prostheses represents several limitations like activation of foreign body reactions or chronic inflammation. Current therapeutic approaches include alloplastic vascular prostheses (Dacron ®, Teflon ®) to replace or bypass the diseased vessel. With a percentage of 46.2%, these diseases represent the largest cause of total deaths. Worldwide, 17.5 million people died of cardiovascular diseases in 2012. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
